
ESS101C
Prof.
Jin-Yi Yu
How PSCs Affect Chlorine?
qIn most of the seasons, there
are always abundant NO2 in the stratosphere to tie up a significant fraction of
the available chlorine in the form of chlorine nitrate.
qIn the
wintertime Antarctic stratosphere, NO2 concentrations are low, because most of the odd nitrogen has
been converted into HNO3 and become droplets in PSCs (polar stratospheric
clouds).
qTherefore, the formation of PSCs
allow reactive chlorine concentration to increases.
qThe PSCs particles also help
convert unreactive forms of chlorine into reactive chlorine by providing surfaces
on which heterogeneous reaction can occur.
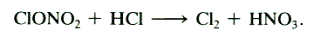
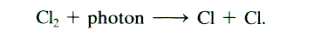

On PCSs
destroy
ozone
